- EN
- ES

An official website of the Biotechnology Innovation Organization
With vaccine development moving so quickly, it is easy to understand why some people are asking whether a vaccine for Covid-19 will be safe and effective. But it is important to remember the very strict scientific and regulatory process vaccine developers must follow to bring a new product to patients, even during the current pandemic. The independent experts at the U.S. Food and Drug Administration (FDA) will not approve a vaccine unless there is data to show the vaccine is:
- Safe for use following a series of random, placebo-based clinical trials of thousands of people;
- Shown to be effective at preventing the disease; and
- Proven to be produced or manufactured consistently, safely and at a high quality.
Visit “How are vaccines developed” to learn more about how the vaccine development process works.
In addition to the FDA’s own rigorous rules and safeguards, there are other layers of oversight to ensure that there is a constant focus on patient safety at every step of the development, approval, and distribution process.
Independent oversight
There are multiple layers of independent oversight built into the existing vaccine development process that are now playing a critical role in the rapid response to the Covid-19 pandemic.
- An Institutional Review Board is comprised of health and scientific experts. According to the FDA, the board’s role is to “assure, both in advance and by periodic review, that appropriate steps are taken to protect the rights and welfare” of individuals enrolled in clinical trials. They have authority to approve or disapprove of a clinical study – or require changes to the research. The review board’s decisions are guided only by what is best for clinical trial participants.
- Data Safety Monitoring Boards are independent groups of experts that include biologists, immunologists, statisticians, and health professionals. They analyze real-time data during the clinical trial process, offering advice and expertise about if and how a trial should move forward or whether one should be paused. These experts provide unbiased evaluation to make sure safety and efficacy rules are being followed and concerns are being addressed along the way.
- The Vaccines and Related Biological Products Advisory Committee is a group within the FDA comprised of 15 independent voting members who are knowledgeable in fields such as immunology, molecular biology, virology, bacteriology, epidemiology, vaccine policy, vaccine safety science, vaccine development, medicine, and infectious diseases, as well as a consumer representative. Following conclusion of a clinical trial, this advisory committee will review the data for the specific vaccine and make a recommendation to the FDA as to whether the product should be authorized or licensed for use.
- The Advisory Committee on Immunization Practices is a group of independent medical and public health experts that advise the Centers for Disease Control and Prevention (CDC). This committee is reviewing safety and efficacy data for clinical trials in real time. Following FDA’s approval of a vaccine, the committee conducts a final review of the safety and efficacy data of the vaccine to make recommendations about who should receive a vaccine and when. They consider things like age, underlying health conditions, and other risk factors to determine the best public use for the vaccine. Visit “When will my family and I receive a vaccine for Covid-19?” to learn more about the ACIP’s pivotal public health role.
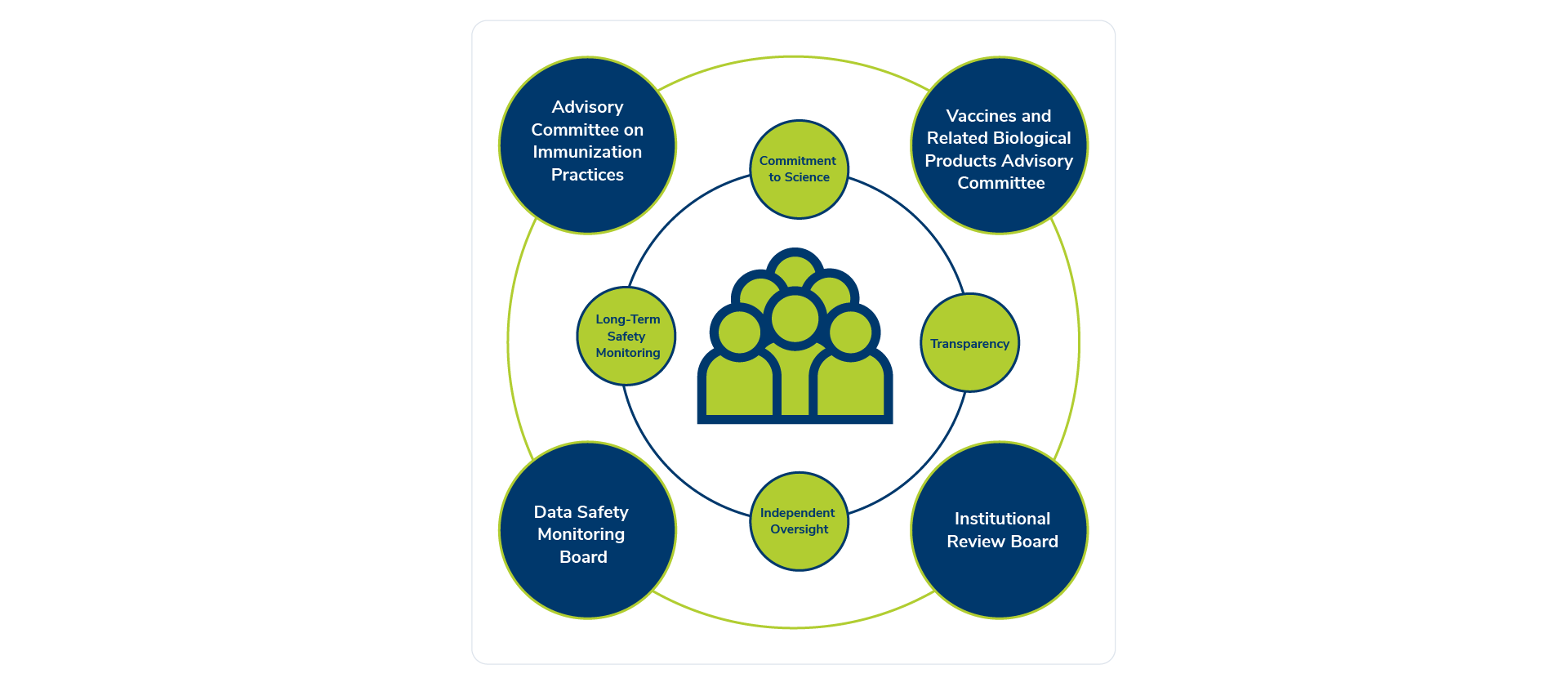
There are multiple layers of independent oversight built into the existing vaccine development process.
Confidence through transparency
Transparency is one of the most important ways to inform the public about – and build greater confidence in – the process to develop a vaccine for Covid-19. Government agencies, like the FDA and CDC, have routinely released key information and held open meetings to allow public feedback to inform their decision-making.
For example, the FDA publicly released guidance that laid out what it expects to see in both safety and efficacy data before approving a Covid-19 vaccine. The FDA also publicly released guidance that explains what researchers will need to show to receive emergency use authorization (see a deeper explanation below) of their vaccines. The public release of both guidance documents provided key insights into the FDA’s decision-making, commitment to transparency, and how researchers are working to develop a vaccine.
Biopharmaceutical research companies have also taken steps to provide greater transparency within the Covid-19 vaccine development process. For instance, it is not uncommon for a pause to take place in clinical trials, but they are rarely reported publicly. However, the decision by several companies to disclose that they had paused clinical trials after safety questions emerged has helped keep the public informed of any bumps in the road to develop a vaccine.
A commitment to science and safety
While there is an urgent need for a Covid-19 vaccine, biopharmaceutical research companies have publicly underscored their firm commitment to letting the science guide the development of a vaccine.
In September 2020, leaders at nine biopharma companies released a joint statement affirming they will “adhere to high scientific and ethical standards” and only submit a vaccine for approval “after demonstrating safety and efficacy” through a Phase 3 clinical study that meets FDA’s strict requirements.
Other leaders within the biotech community released an open letter urging everyone working against Covid-19 to ensure the “integrity, transparency, and objective assessment” of clinical data in order to promote the public’s trust in any treatment intended to help address the pandemic.
Safety is the priority, even in an emergency
There has been a lot of discussion about whether the FDA should use its authority to grant what is known as “emergency use authorization” for a Covid-19 vaccine and what this might mean for patient safety. The FDA has granted these types of authorizations before, but typically it approves a vaccine through a Biologic License Authorization, which requires years of clinical data for what is considered full FDA approval.
Following multiple public health threats, in 2004, the FDA was given the legal authority to allow the “emergency use” of medical products that had not received full FDA approval. The idea was that during a public health crisis, like the current pandemic, the benefits to families and communities outweighed the known risks of allowing the use of a medical product before it had been approved.
During a public health emergency, the FDA still sets an extremely high bar for emergency use of any vaccine, and that is true in the case of the current pandemic. Also, any emergency use is limited to those at higher risk of infection or serious disease. This ensures we prioritize protecting the most vulnerable populations – like the elderly and our frontline health care workers – while continuing to gather data for broader public use.
If you want to learn more about how emergency use works and what it means for patients, be sure to watch this short, informative video developed by the experts at the FDA.
Long-term safety monitoring
Even after a Covid-19 vaccine is approved, the FDA, CDC, healthcare systems and vaccine developers will continue to monitor the safety and efficacy of the vaccine for years. Covid-19 vaccines will be subject to the same long-term safety requirements and will be utilizing the same safety monitoring systems as routine vaccines. Meanwhile, the CDC and FDA are creating a new monitoring system, VSAFE, that will allow individuals to submit any safety issues via text message.
Health care providers are an additional layer of protection at the state and local levels. This includes people like your physician, pharmacists, and state health departments. These health professionals are required to report any adverse effects – however rare – to the FDA and the CDC.
The FDA’s multi-tiered safety systems, along with vaccine developers and local health care providers, ensure that no one cuts corners when it comes to patient safety.